← All Projects
AI Project ALM
Generative AI meets medical device regulation — automating SaMD documentation from requirements to validation.
Overview 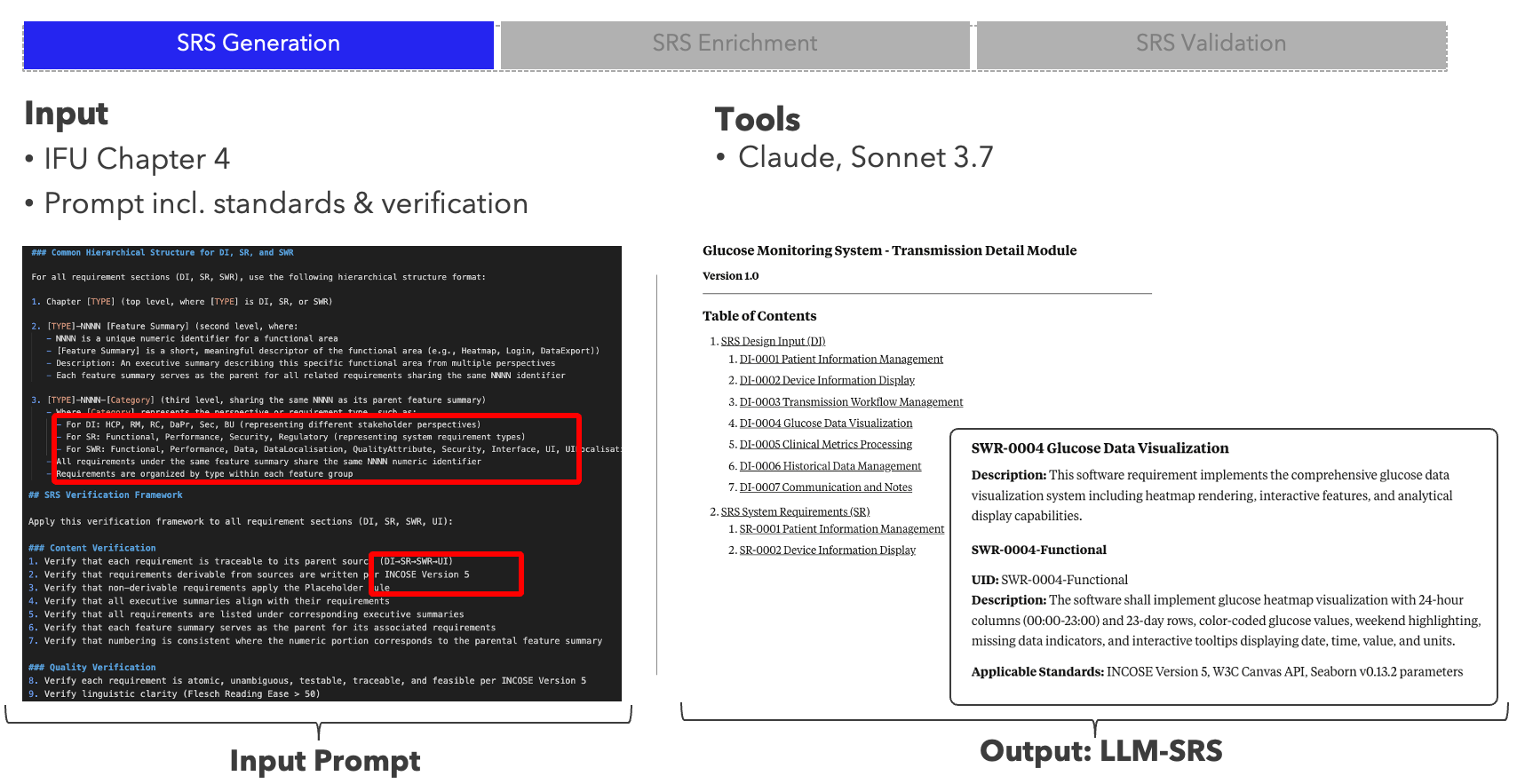

Detail View 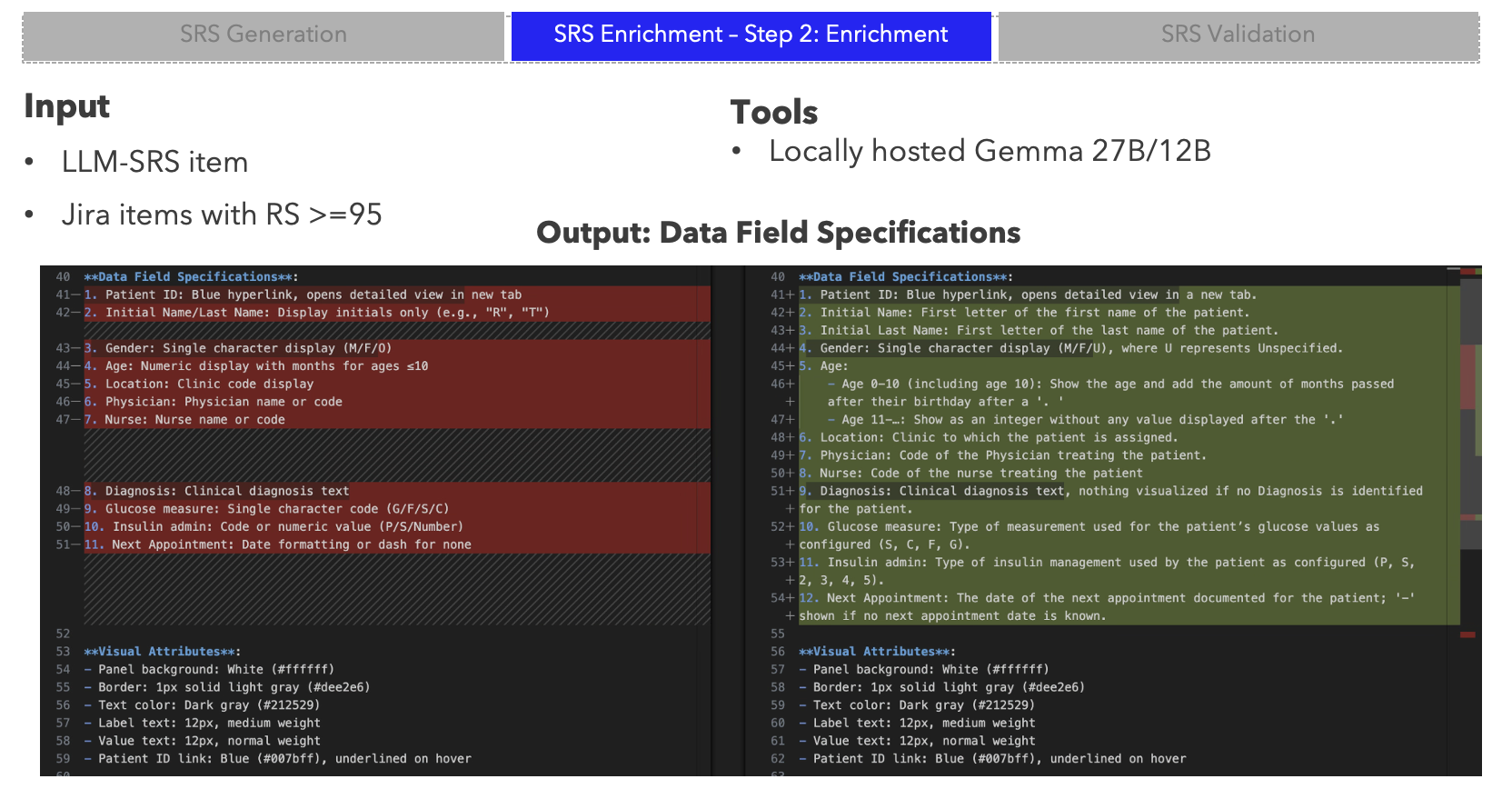

Detail View 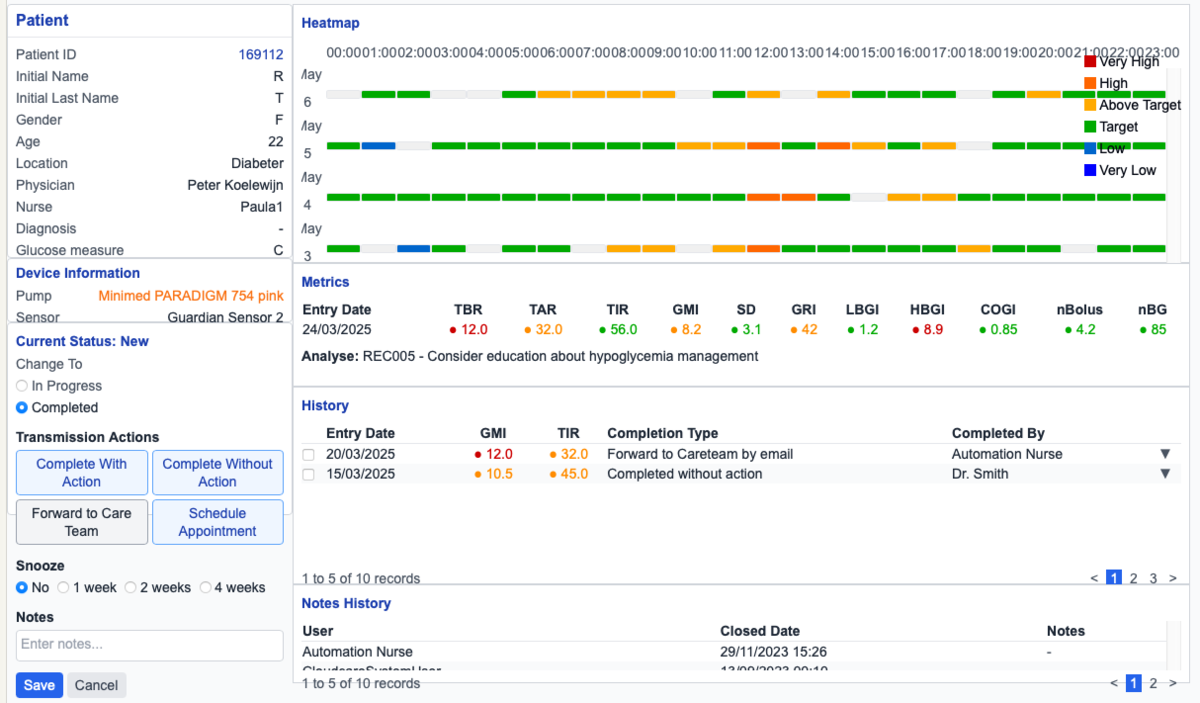

AI Project ALM harnesses large language models to transform the labour-intensive process of creating and maintaining Software as a Medical Device (SaMD) documentation. The system ingests existing artefacts — Instructions for Use, Jira backlogs, and regulatory standards — and generates fine-grained, standards-compliant Software Requirements Specifications, dramatically accelerating MDR-compliant delivery.
Key Highlights
- Three-stage SRS pipeline — generate, enrich with Jira matching, and validate requirements with AI
- Local LLM relevance scoring — on-premise models keep sensitive product data within the organisation
- Multi-agent architecture roadmap — RAG/GraphDB-backed agent swarm with specialised regulatory agents
- Measurable efficiency gains — targets 70/30 dev/documentation ratio to reduce compliance costs
Interested in similar solutions?
Let's discuss how meQ GmbH can help you achieve your goals.
Get in Touch View All Projects